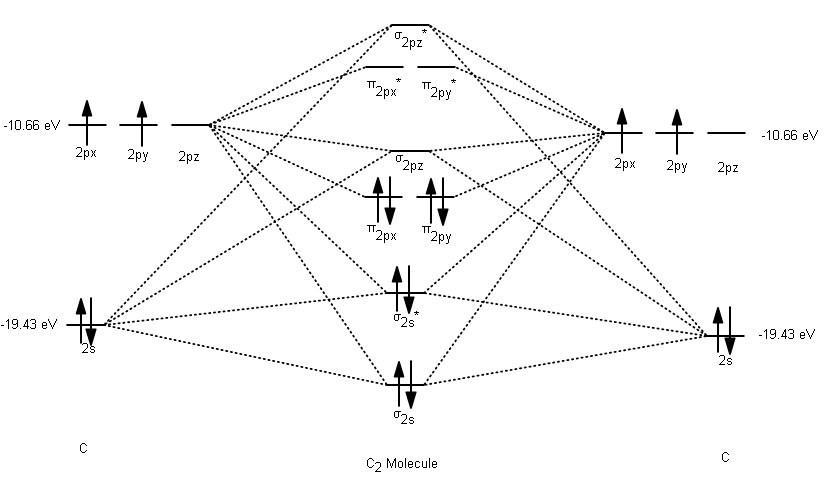 
With which of the lithium orbitals does the hydrogen 1 s orbital interact? The lithium 1 s orbital is the lowest-energy orbital on the diagram. There are two occupied atomic orbitals on the lithium atom, and only one on the hydrogen. One thing that makes this diagram look different from the ones we have seen previously is that the parent atomic orbitals have widely differing energies the greater nuclear charge of lithium reduces the energy of its 1 s orbital to a value well below that of the 1 s hydrogen orbital. The diagram shows how the molecular orbitals in lithium hydride can be related to the atomic orbitals of the parent atoms. Lithium hydride is a stable, though highly reactive molecule. As an example of a heteronuclear molecule, let’s take a look at a very simple example- lithium hydride. 084 kJ/mol this is not enough to hold the two atoms together in the presence of random thermal motion at ordinary temperatures, so dihelium dissociates as quickly as it is formed, and is therefore not a distinct chemical species.Īll the molecules we have considered thus far are homonuclear they are made up of one kind of atom. Experimentally, the bond energy of dihelium is only. The one orbital almost exactly cancels out the effect of the other. You should now be able to predict that He 2 cannot be a stable molecule the reason, of course, is that we now have four electrons- two in the bonding orbital, and two in the antibonding orbital. Taking our building-up process one step further, we can look at the possibilities of combining to helium atoms to form dihelium. The presence of an electron in this orbital, as we have seen, gives rise to a repulsive component which acts against, and partially cancels out, the attractive effect of the filled bonding orbital. The reason for this should be obvious two electrons were accommodated in the bonding orbital, but the third electron must go into the next higher slot- which turns out to be the sigma antibonding orbital. This molecule is stable, but not as stable as dihydrogen the energy required to break He 2+ is 301 kJ/mole. We can think of it as containing two helium nuclei and three electrons. The dihelium positive ion is a three-electron molecule. With two electrons we are still ahead, so let’s try for three. 
Although H 2 + is stable in this energetic sense, it happens to be an extremely reactive molecule- so much so that it even reacts with itself, so these ions are not commonly encountered in everyday chemistry. This single electron is nevertheless enough to lower the potential energy of one mole of hydrogen nuclei pairs by 270 kJ- quite enough to make them stick together and behave like a distinct molecular species. Since any orbital can hold a maximum of two electrons, the bonding orbital in H 2 +is only half-full. Thus, the single electron in this simplest of all molecules goes into the bonding orbital, leaving the antibonding orbital empty. Atomic valence electrons (shown in boxes on the left and right) fill the lower-energy molecular orbitals before the higher ones, just as is the case for atomic orbitals. This scheme of bonding and antibonding orbitals is usually depicted by a molecular orbital diagram such as the one shown here for the dihydrogen ion H 2 +.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
